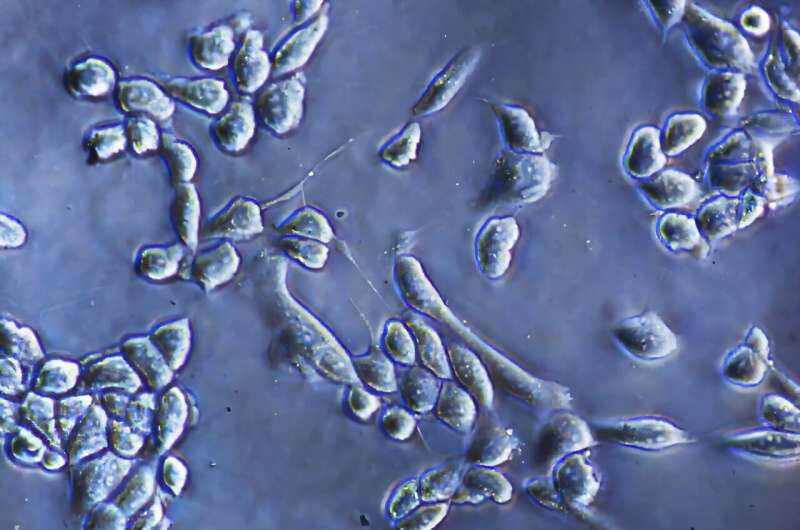
Researchers at the Massachusetts Institute of Technology (MIT) have developed a novel enzyme-free method for detaching cells from culture surfaces, a significant advancement for the biomedical field. This new technique, detailed in a paper published in the journal ACS Nano, utilizes alternating electrochemical currents on a specialized biocompatible polymer nanocomposite to release cells while maintaining over 90 percent cell viability.
Traditionally, the detachment of anchorage-dependent cells—those that require a solid surface to thrive—relies on enzymatic methods that can compromise cell integrity. According to Kripa Varanasi, a professor of mechanical engineering at MIT, these enzymatic treatments pose several challenges. They can damage delicate cell membranes and surface proteins, particularly in primary cells, and often involve complex, multistep processes that slow down workflows. Moreover, the reliance on enzymes derived from animals raises compatibility concerns for applications in human therapies.
Innovative Electrochemical Technique
The enzyme-free approach not only simplifies the detachment process but also has the potential to transform biomanufacturing practices. By applying low-frequency alternating voltage, the new platform disrupts cell adhesion within minutes. This method significantly reduces the common issues associated with enzymatic and mechanical techniques, such as cell damage and excessive waste generation. Current estimates suggest that enzymatic methods contribute to approximately 300 million liters of cell culture waste each year.
The researchers’ innovative technique allows for uniform application across large areas, making it ideal for high-throughput and large-scale applications like cell therapy manufacturing. Bert Vandereydt, a co-first author and mechanical engineering researcher at MIT, emphasizes the method’s industrial scalability, envisioning fully automated, closed-loop cell culture systems in the near future.
Applications in Medicine and Biomanufacturing
The impact of this new technique extends beyond routine cell culture. It opens pathways for the safe expansion and harvesting of sensitive immune cells, particularly for applications in therapies such as CAR-T, a form of immunotherapy targeting cancer cells. The platform’s ability to dynamically shape the ionic microenvironment around cells introduces opportunities for enhanced control over cellular processes, including ion channel regulation and signaling pathway studies.
Wang Hee (Wren) Lee, a postdoctoral researcher at MIT and co-first author of the study, highlighted the dual significance of their work. “Our research demonstrates how electrochemistry can be harnessed not just for scientific discovery, but also for scalable, real-world applications,” he stated. The implications for reducing waste and accelerating automation in biomanufacturing are substantial.
Collaboration with the Broad Institute has further underscored the biomedical significance of the research. Yuen-Yi (Moony) Tseng, a principal investigator at the Broad Institute, noted the method’s potential to streamline workflows in research and clinical biomanufacturing, ultimately preserving cell functionality for therapeutic use.
In experimentation, the team tested their method on human cancer cells, including those from osteosarcoma and ovarian cancer. The results were promising; the detachment efficiency increased dramatically from 1 percent to 95 percent, with cell viability remaining above 90 percent.
This groundbreaking enzyme-free method illustrates a significant shift in cell culture techniques, paving the way for advancements in biotechnology, pharmaceuticals, and regenerative medicine. As researchers continue to refine and implement this technology, the potential for improved efficiency and reduced environmental impact in cell culture practices becomes increasingly tangible.