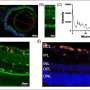
A groundbreaking study published in Nature Communications reveals that cancer cells possess a distinctive nuclear metabolic fingerprint. Researchers discovered that more than 200 metabolic enzymes, typically involved in energy production within mitochondria, are also located directly on human DNA.
This research indicates that various types of cells, tissues, and cancers exhibit unique patterns of these enzymes, which are compartmentalized within the cell nucleus. This finding offers the first evidence of a metabolic interaction between enzymes and DNA, a phenomenon the authors describe as a “nuclear metabolic fingerprint.”
The implications of this discovery could extend beyond basic science. Understanding how these enzymes interact with DNA might pave the way for innovative cancer treatments. The identification of specific metabolic signatures could lead to more personalized therapies, targeting the unique characteristics of different tumors.
Researchers employed advanced techniques to analyze the distribution of these enzymes in various cell types. They found that the presence and arrangement of these metabolic enzymes differ significantly between healthy cells and cancerous ones. This variance could provide critical insights into how cancer cells manage energy differently, potentially fueling their rapid growth and resistance to conventional therapies.
This innovative research opens new avenues for exploring how metabolic processes influence cellular behavior. It challenges the long-held view that metabolic functions are confined primarily to mitochondria, emphasizing the need for further investigations into nuclear metabolism.
Future studies will likely focus on the potential of targeting these nuclear metabolic pathways as a therapeutic strategy. By disrupting the unique metabolic profiles of cancer cells, researchers hope to inhibit their growth and improve treatment outcomes.
In summary, the identification of a nuclear metabolic fingerprint marks a significant advancement in cancer research. This finding could lead to breakthroughs in understanding tumor biology and developing new therapeutic approaches tailored to individual patients.